The National Coalition of STD Directors, in partnership with Chembio, is excited to present an informational webinar featuring different presenters from across the nation who will share their experiences using the DPP® HIV-Syphilis Tests in the ongoing HIV and syphilis epidemics.
Date: May 27th, 2021
Time: 2:30pm-3:45pm EST
Host: National Coalition of STD Directors
The recently released 2019 STD Surveillance Report revealed there were over 129,000 cases of syphilis, showing an increase of more than 70 percent since 2015. We also know that the risk of acquiring an HIV infection is increased when syphilis is present. It’s important now more than ever for those at the frontlines of these epidemics to have as many tools in their STD prevention toolbox as possible.
We hope you will join us in hearing diverse examples of how this innovative tool has been utilized!
Speakers
• Kayla Wilburn, Clinical Director – CAPSLO, San Luis Obispo, CA
• Russell R. Rooms, MSN, APRN-CNP – Diversity Family Health, Oklahoma City, OK
• Johnny Gonzalez, MD, Program Director – KC Care Health Centers, Kansas City, MO
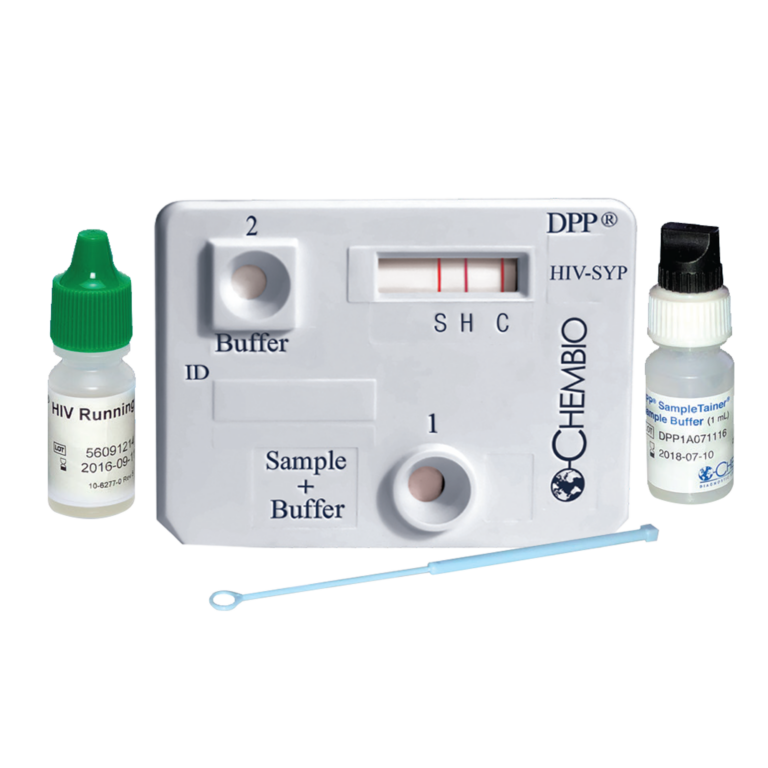
Chembio DPP® HIV-Syphilis Assay is a dual rapid test for the detection of antibodies to HIV 1/2 and Treponema pallidum in fingerstick whole blood, venous whole blood, serum or plasma specimens.
Chembio’s proprietary DPP® technology differs from classical lateral flow tests by operating in a manner similar to that of the sequential ELISA format which is not sensitive to the “Hook Effect”.
Highlights:
• Patented DPP® technology allows for higher sensitivity and specificity
• 2 test results in a single sample
• Small sample volume: 10 μl
• DPP sample tainer included for safe and closed sample handling
• CE Marked, FDA approved