RESZON® LYMPHATIC FILARIASIS RAPID TEST KIT
BRUGIA RAPID
This webpage content is intended for Healthcare Professionals only, not for general public.
Registered under Act 737
MDA registration number: IVDC8841920-38490
- Rapid immunochromatography assay for brugian filariasis infection
- Detect specific IgG4 antibodies to Brugia spp. (Brugia malayi and Brugia timori)
- Sample collected at any time of the day can be used
- Simple to perform & no special equipment required
SPECIFICATION
| Platform | Immunochromatographic Assay |
| Format | Cassette |
| Detection | IgG4 antibodies specific to Brugia spp. (Brugia malayi and Brugia timori) |
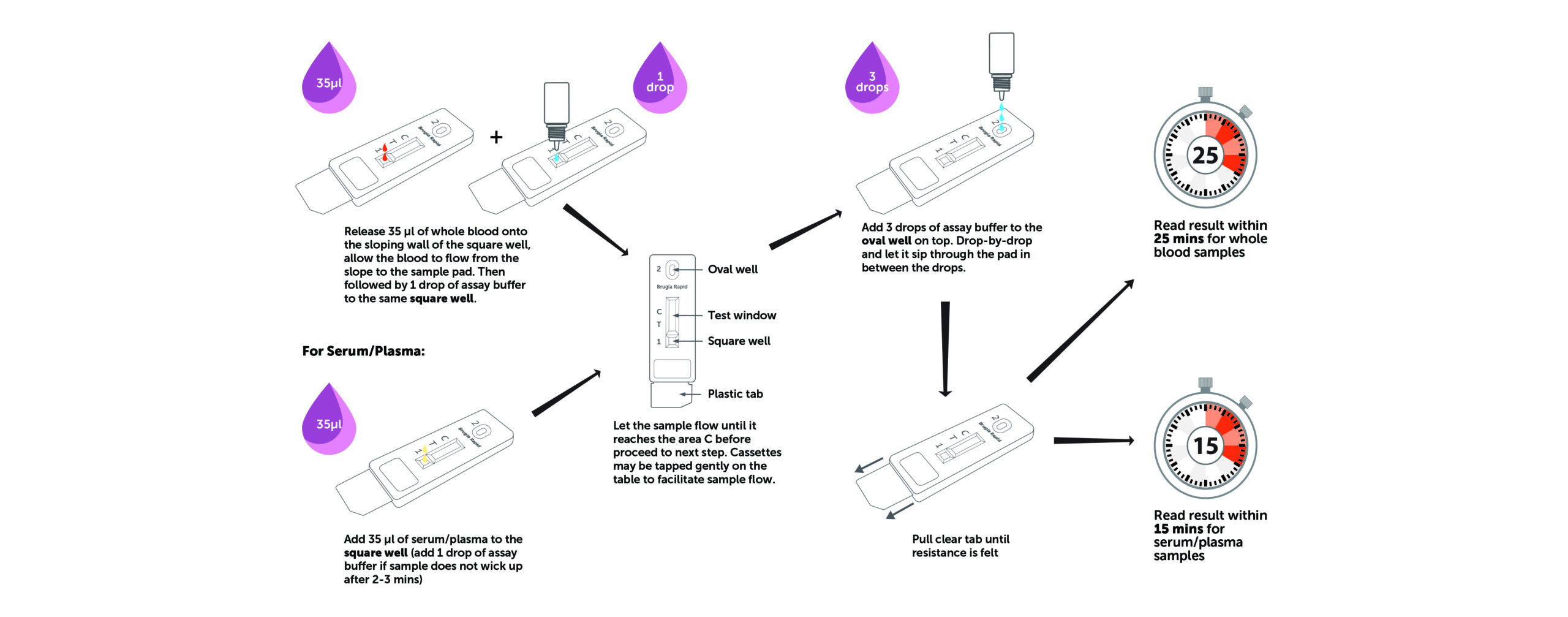
| Specimen | Human whole blood, serum or plasma |
| Specimen Volume | 35 µl |
| Sensitivity | 94.75% |
| Specificity | 99.26% |
| Storage | 4-30°C |
| Assay Time | 15-25 minutes |
| Shelf Life* | 24 months |
*from date of manufacture
|
Cat. No. |
Product Name |
Detection |
Packing Size |
|---|---|---|---|
|
RLF-RD0201 |
RESZON® Lymphatic Filariasis Rapid Test Kit-Brugia Rapid |
Antibodies specific to B. malayi and/or B. timori |
25 T/ Kit |
Frequently Asked Questions (FAQ) for RESZON® Lymphatic Filariasis Rapid Test Kit Brugia Rapid
Before running the test
1. Can I use the test device if the pouch is damaged or there is broken/ incomplete seal?
No. Do not use the test device if the pouch is damaged or the seal is broken/ incomplete. Please use a new test.
2. Can I use the test device if there is no desiccant in the pouch?
No. Desiccant should come together with test device in the pouch. Please use a new test.
3. Can I use the kit beyond its expiry date?
No. User should not use the test beyond the expiry date shown on the test kit labelling.
While running the test
4. Can I use specimen that has been stored in refrigerator?
No. Refrigerated specimen should be equilibrated to room temperature (about 30 minutes) or between 15°C and 30°C before being tested. Remember to mix the specimen before use.
5. Can I use haemolyzed specimen?
No. Haemolyzed specimen cannot be used on this test. Haemolyzed samples may interfere with test results.
6. Can I use whole blood sample from finger prick?
Yes. Whole blood sample from finger prick can be used. However, skillful professional user and a suitable lancet are required to ensure suffi cient specimen can be collected. It is highly recommended to use whole blood sample from venipuncture.
7. I found that the buffer is not enough to run the test in the kit. Can I use bu ffer from other brand?
No. Buffer from other brand cannot be used as a substitute for the buffer in this kit. Please contact your supplier for assistance.
The buffer provided in the kit are more than the actual volume needed to run all 25 tests in the kit. User should check the IFU again and ensure that the tests are run following the correct assay procedure.
8. Can I put more sample to the test? What if I accidentally added more sample to the test?
No. User should follow the instruction of the assay procedure strictly. Failure to follow the instruction may give inaccurate test results. Repeat the test if the sample volume is incorrect during the testing.
9. Why is it the sample never flow up, or the sample only flow half of the test window?
It may be due to insufficient specimen or buffer volume. Sufficient amount of specimen and buffer are important to ensure the sample to flow up properly. Please repeat the test with a new test device if invalid results are observed.
When adding buffer, it is important to hold the buffer bottle vertically, carefully press the bottle to release full drop of buffer, one drop by one drop. The droplet volume of the buffer may be inconsistent if the buffer bottle is not held vertically above the well “S”, or buffer being released too fast.
10. Can we add more buffer if the sample is not flowing up?
No. The assay procedures need to be followed strictly. If no sample flow is observed, check again the procedure and repeat the test with a new test device.
11. Can I read results before 20 minutes?
The reading time depends on the type of sample used. For whole blood sample, results can be read within 25 minutes. For serum/plasma sample, read the results within 15 minutes.
12. Can I read results after 25 minutes?
The reading time depends on the type of sample used. For whole blood sample, results can be read within 25 minutes. For serum/plasma sample, read the results within 15 minutes.
Interpretation of results
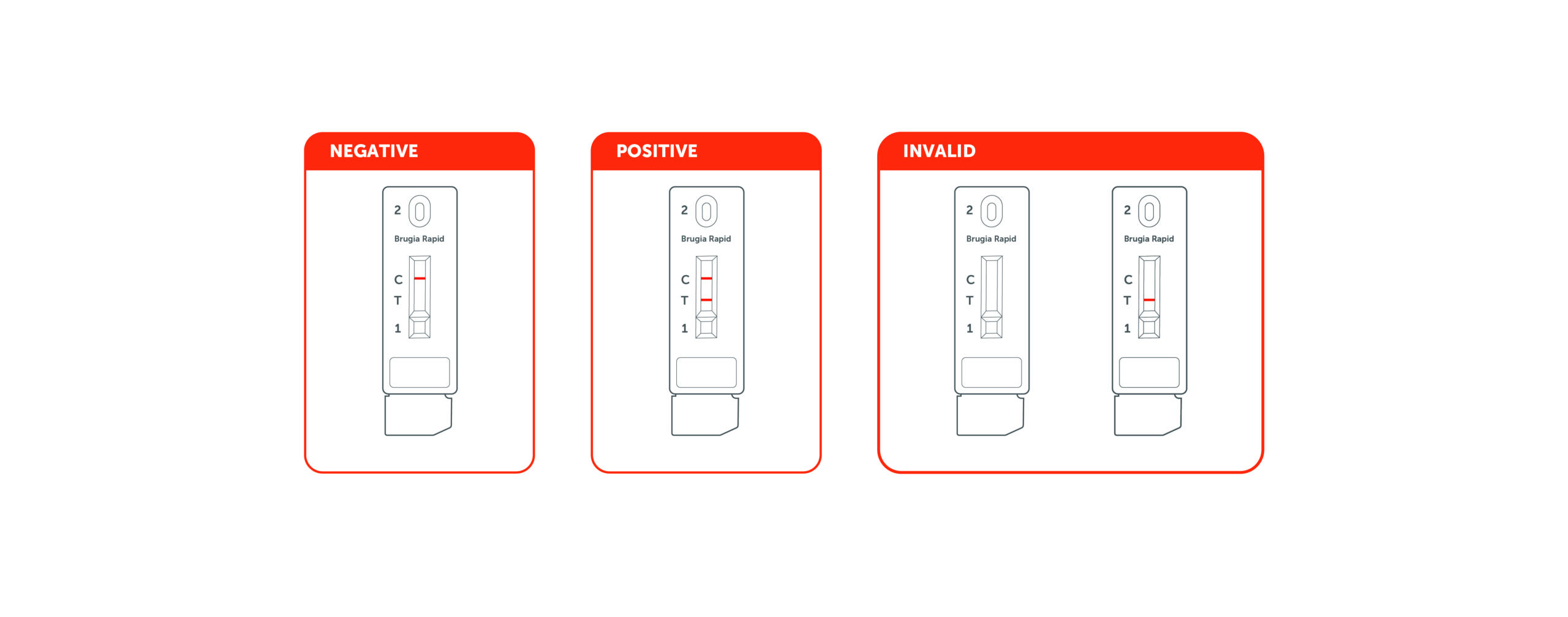
13. What do the results mean?
If coloured bands appear at the control line (C) and test line (T), it indicates a positive result. Positive results indicate the presence of either Brugia malayi and/or Brugia timori specific IgG4 antibodies in the specimen. Positive result should not be used as the sole criteria for the diagnosis of filariasis infection. Positives results should be confirmed by alternative testing method and clinical finding before positive determination is made.
The result is negative if only control line (C) is visible with no coloured line developed at test line (T) position. This indicates that Brugia malayi and/or Brugia timori specific IgG4 antibody has not been detected. Negative result does not preclude the possibility of exposure to or filariasis infection. It is recommended that the results obtained be interpreted in conjunction with other diagnostic results and clinical information.
14. Can the test identify or di erentiate between the two target species?
The test can detect either Brugia malayi and/or Brugia timori specific IgG4 antibodies in the specimen if present, but cannot identify or differentiate between the species.
15. What to do if the results showed negative result, but the symptoms persist?
If the symptom persists, while the result is negative or non-reactive result, it is recommended to re-sample the patient a few days later or use alternative testing such as blood smear or ELISA.
After running the test
16. What should I do after running the test?
Handle all materials that come in contact with patient samples as infectious waste and dispose in a biohazard container.
Good Laboratory Practice
17. How is the good quality control for RESZON® Brugia Rapid Test?
Users should follow respective Good Laboratory Practice SOP of the labs. However, it is recommended to use external controls, positive and negative, to confirm the test procedure and to verify proper test performance, particularly under the following circumstances:
- New operator uses the kit, prior to performing testing of specimens.
- A new lot of test kits is used.
- A new shipment of test kits is used.
- The temperature used during storage of the kit falls outside of 4-30°C.
- The temperature of the test area falls outside of 15-30°C.
- To verify a higher-than-expected frequency of positive or negative results.
- To investigate the cause of repeated invalid results.






