RESZON® DOA RAPID CUP TEST
This webpage content is intended for Healthcare Professionals only, not for general public.
Registered under Act 737
MDA registration no. IVDB10354125-196558
- Rapid, competitive binding immunoassay for qualitative determination of different drugs and drugs metabolites in human urine
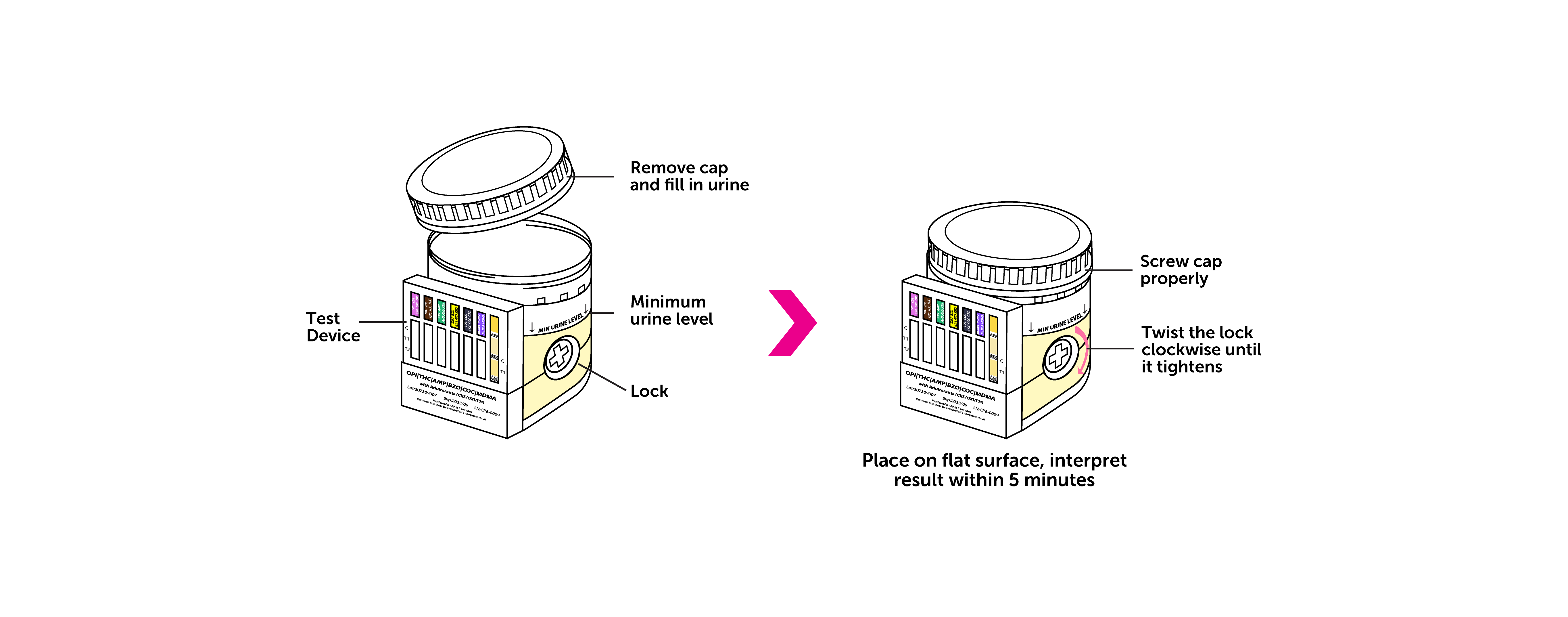
- Use a self-contained, one-step closed system with a secure lid to ensure total integrity of urine specimen
- Comes with integrated key activator (no dipping or pouring required)
- Able to detect from 4 to 12 different drug combinations at the same time
- Easy result interpretation as each drug type is clearly labeled and color-coded
| Platform | Immunochromatographic Assay |
| Format | Cup/ Multiplex cup |
| Specimen | Human urine |
| Assay time | 5 minutes |
| Shelf Life* | 24 months |
|
Cat. No. |
Product Description |
Test Specimen |
Test Format |
Packing Size |
|---|---|---|---|---|
|
RDT-RDC04 |
RESZON® DOA Rapid, Cup 4-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC05 |
RESZON® DOA Rapid, Cup 5-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC06 |
RESZON® DOA Rapid, Cup 6-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC07 |
RESZON® DOA Rapid, Cup 7-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC10 |
RESZON® DOA Rapid, Multiplex Cup 10-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC12 |
RESZON® DOA Rapid, Multiplex Cup 12-in-1 |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC04-AD1 |
RESZON® DOA Rapid, Cup 4-in-1, with adulterant tests (CRE, OXI, pH) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC04-AD2 |
RESZON® DOA Rapid, Cup 4-in-1, with adulterant tests (SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC04-AD3 |
RESZON® DOA Rapid, Cup 4-in-1, with adulterant tests (CRE, OXI, pH, SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC05-AD1 |
RESZON® DOA Rapid, Cup 5-in-1, with adulterant tests (CRE, OXI, pH) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC05-AD2 |
RESZON® DOA Rapid, Cup 5-in-1, with adulterant tests (SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC05-AD2 |
RESZON® DOA Rapid, Cup 5-in-1, with adulterant tests (SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC05-AD3 |
RESZON® DOA Rapid, Cup 5-in-1, with adulterant tests (CRE, OXI, pH, SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC06-AD1 |
RESZON® DOA Rapid, Cup 6-in-1, with adulterant tests (CRE, OXI, pH) |
Urine |
Cup |
1 T/ Kit |
|
RDT-RDC06-AD2 |
RESZON® DOA Rapid, Cup 6-in-1, with adulterant tests (SG, NIT, GLU) |
Urine |
Cup |
1 T/ Kit |
Frequently Asked Questions (FAQ)
Before running the test
1. Can I use the test device if the pouch is damaged or there is broken/ incomplete seal?
No. Do not use the test device if the pouch is damaged or the seal is broken/ incomplete. Please use a new test.
2. Can I use the test device if there is no desiccant in the pouch?
No. Desiccant should come together with test device in the pouch. Please use a new test.
3. Can I use the kit beyond its expiry date?
No. User should not use the test beyond the expiry date shown on the test kit labelling.
4. Can medications a ect the results?
Yes. Certain prescription medicines may cause cross-reactions and lead to false positives. DOA rapid tests are only screening tools. Any positive result should be confirmed by a laboratory method such as GC-MS (Gas Chromatography–Mass Spectrometry) or LC-MS/MS.
5. Can the test show how much drug is in the system?
No. The test is qualitative test not a quantitative test. The test only indicates whether the concentration is above or below the cut-o level, not the exact amount.
While running the test
6. Besides human urine, can this test be used to test animal urine?
No. Only use human urine sample.
7. Can I use refrigerated urine sample?
Urine specimens may be stored at 2-8°C for up to 48 hours prior to testing. For prolonged storage, specimens may be frozen and stored below -20°C. Frozen specimens should be thawed and mixed well before testing. However, it is recommended to use fresh urine sample whenever possible.
8. What if the urine sample has precipitates?
Urine specimens exhibiting visible precipitates should be allowed to settle to obtain a clear specimen for testing.
9. Why is it the sample never flow up, or the sample only flow half of the test window?
It may be due to insufficient specimen volume. Sufficient amount of specimen is important to ensure the sample to flow up properly. Please check if the lock has been twisted completely. Incomplete twist of the lock may prevent enough specimen from entering the test device well. Repeat the test with a new test device if invalid results observed. Make sure the specimen volume reaches the minimum level indicated on the cup.
10. How should I interpret the adulterant test results?
For the device that comes with adulterant test, read the adulterant test result within 1-2 minutes by comparing the reaction area to the colour chart provided. Hold the cup side by side for comparison and match carefully.
11. Can I read results before 5 minutes?
Results must be interpreted within 5 minutes.
12. Can I read results after 5 minutes?
No. Do not read results after 5 minutes. Results read after 5 minutes may lead to a false positive, false negative, or invalid result.
Interpretation of results
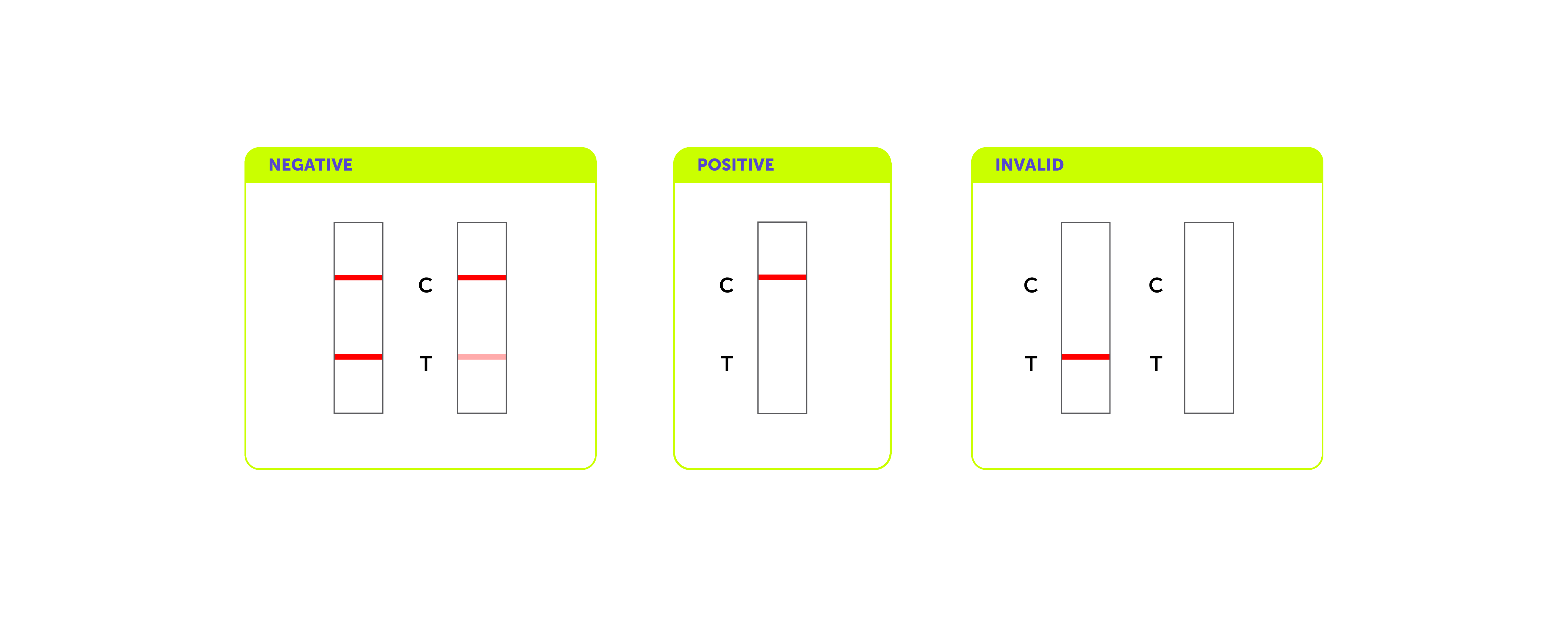
13. What do the results mean?
A purplish-pink color band appears in the control zone but not in the test zone. This is a positive result and indicates the drug level is at or above the detection sensitivity. It is recommended that all positive results to be confirmed by using an appropriate analytical
technique.
The result is negative if both control line (C) and test line (T) are visible. This indicates that the drug concentration is below the detection sensitivity.
If no bands appear, or a test band appears without a control band, disregard the results. The presence of a control line is necessary to validate test performance. If this occurs, the assay should be repeated with a new test device.
What should I do after running the test?
Urine specimens may be infectious, properly handle and dispose of all used reaction devices into a biohazard container.
How is the good quality control for RESZON® DOA Cup?
Users should follow respective Good Laboratory Practice SOP of the labs. However, it is recommended to use external controls, positive and negative, to confirm the test procedure and to verify proper test performance, particularly under the following circumstances:
- New operator uses the kit, prior to performing testing of specimens.
- A new lot of test kits is used.
- A new shipment of test kits is used.
- The temperature used during storage of the kit falls outside of 4-30°C.
- The temperature of the test area falls outside of 15-30°C.
- To verify a higher-than-expected frequency of positive or negative results.
- To investigate the cause of repeated invalid results.
After running the test
14. What should I do after running the test?
Urine specimens may be infectious, properly handle and dispose of all used reaction devices into a biohazard container.
Good Laboratory Practice
15. How is the good quality control for RESZON® DOA Cup?
Users should follow respective Good Laboratory Practice SOP of the labs. However, it is recommended to use external controls, positive and negative, to confirm the test procedure and to verify proper test performance, particularly under the following circumstances:
- New operator uses the kit, prior to performing testing of specimens.
- A new lot of test kits is used.
- A new shipment of test kits is used.
- The temperature used during storage of the kit falls outside of 4-30°C.
- The temperature of the test area falls outside of 15-30°C.
- To verify a higher-than-expected frequency of positive or negative results.
- To investigate the cause of repeated invalid results.


