12 Oct 2022
Reszon Diagnostics International Sdn Bhd (Reszon) today announced its launch of commercial distribution of Chembio HIV rapid point-of-care assays.
HIV 1/2 STAT-PAK® Assay is a WHO prequalified, rapid point-of-care assay for the detection of HIV-1 and HIV-2 antibodies in fingerstick whole blood, venous whole blood, serum or plasma. It is the only HIV rapid test that exceed WHO performance thresholds with 100% sensitivity and 100% specificity.
In comparison with HIV 1/2 STAT-PAK® Assay, SURE CHECK® HIV 1/2 Assay is designed with unique all-in-one barrel sample collection and test device bundled with safety lancet and bandage, giving the healthcare professionals a piece of mind and enhanced convenience to perform the test. SURE CHECK® HIV 1/2 Assay is a WHO Prequalified rapid point-of-care test that detects antibodies to HIV-1 and HIV-2 in fingerstick whole blood, venous whole blood, serum and plasma specimens.
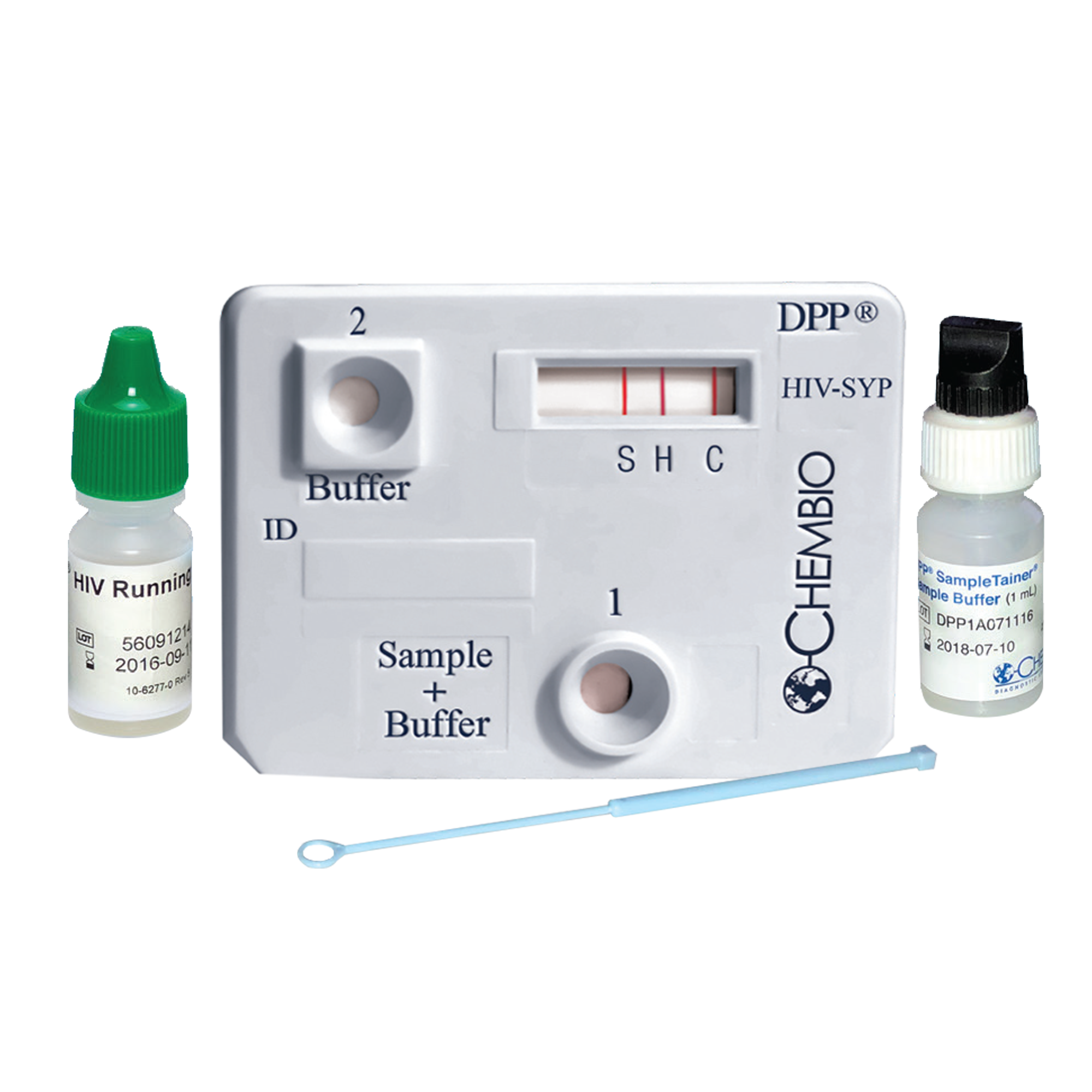
DPP® HIV-Syphilis Assay is the first and only FDA-approved HIV-Syphilis dual rapid test for the detection of antibodies to HIV 1/2 and Treponema pallidum in fingerstick whole blood, venous whole blood, serum, or plasma specimens. The test device is designed with patented DPP® technology – the Chembio’s propriety technology that differs from classical lateral flow tests by operating in a manner similar to that of the sequential ELISA format which is not sensitive to the “Hook Effect”, allowing significant increase of analytical and clinical sensitivity.
“We are excited about the addition of Chembio’s HIV rapid point-of-care assays into our menu of infectious diseases rapid test kit range,” said Mr. EL Law, Chief Executive Officer of Reszon.
“We are now ready to offering customer reliable and highly qualified HIV rapid testing solution for local and South East Asia export markets.”