The world commemorates World AIDS Day on 1st Dec each year.
It’s an opportunity for people worldwide to unite in the fight against HIV, to show support for people living with HIV, and to commemorate those who have died from an AIDS-related illness.
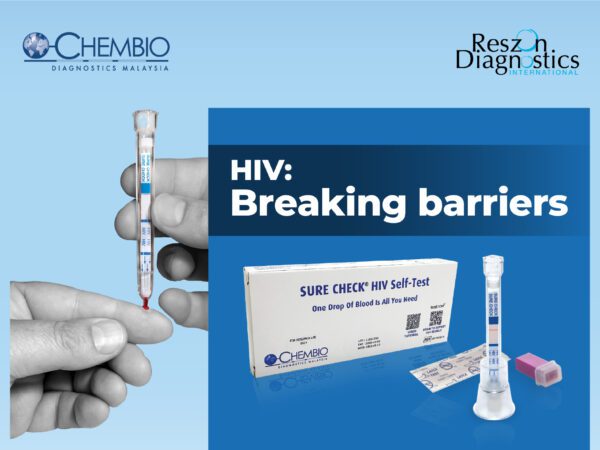
Rapid point-of-care assay is the key medical diagnostic tool to detect presence of HIV-1 and HIV-2 antibodies in patient’s specimen and helps to avoid transmission among the community. Rapid HIV screening empowers medical professionals to make early and clinical diagnosis in fast and accurate manner even in remote areas.
Why choose Chembio HIV 1/2 STAT-PAK® Assay manufactured by Reszon?
- Produced in accordance with World Health Organization Pre-qualified facility
- Reliable standards: FDA approved, CE marked, WHO prequalified
- Affordable pricing to enable more testing
We are actively looking for distributors in South East Asia to market Chembio’s HIV testing solutions, visit our website or contact Reszon’s sales team to find out more.