-

Reszon at Analitica Latin America 2017 and Visit to HCDMUSP
-Reszon Diagnostics International Sdn Bhd (Reszon) showcased at Analitica Latin America 2017, São Paulo Expo, Brazil from 26-28 September 2017. Reszon’s Drug of Abuse Rapid Tests were being highlighted and featured during the expo.
00 -
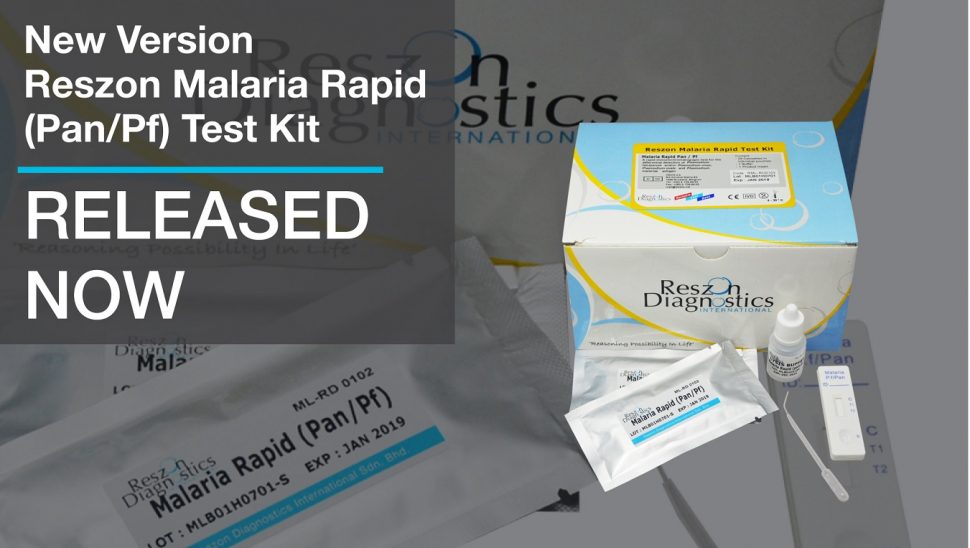
New Version of Reszon Malaria Rapid (Pan/Pf) Test Kit – Released Now
-We are proud to announce that the new version of Malaria Rapid (Pan/Pf) Test is now available at Reszon! Malaria Rapid (Pan/Pf) test is a rapid immunochromatography assay used for qualitative detection and differentiation of Plasmodium falciparum and/or Plasmodium vivax, Plasmodium ovale and Plasmodium malariae antigen in whole blood.
-

Product Training and Demonstration of PanLF and Brugia Rapid Test for Transmission Assessment Survey (TAS) Program for Ministry of Health, Malaysia
-On 10th July 2017, Reszon Diagnostics International Sdn Bhd (Reszon) was invited by the Ministry of Health Malaysia to conduct product training and demonstration of PanLF Rapid Test and Brugia Rapid Test.
-

Factory Visit by Ministry of Defence Malaysia (MINDEF), 6 Sep 2017
-On 6th Sept 2017, Reszon Diganostics International Sdn Bhd (Reszon) was honored to host a prestigious visit for officers from the Ministry of Defence, Malaysia (MINDEF). Officers from 2 main wings of MINDEF i.e. the Malaysian Army and the Royal Malaysian Navy were taken a tour around our manufacturing facility, especially the production line of urine drug tests.
-

PanLF Rapid Test and Brugia Rapid Test in GPELF Program
-The Global Program to Eliminate Lymphatic Filariasis (GPELF) was initiated by WHO since year 2000 with the ultimate objectives to stop the spread of filariasis infection and to control morbidity through continuous collaborative effort worldwide.
Reszon News
Reszonics > Reszon News
